Polymer Stabilised Nanoparticles
Nanoparticles can often exhibit a diverse range of properties that significantly differ from the bulk materials. For gold nanoparticles (AuNPs), some illustrative examples of these interesting properties include:
- strong absorption band in the visible region,1
- surface-enhanced Raman scattering (SERS) of adsorbed molecules,2-4
- catalytic behavior,5-9
- quenching of proximal fluorophores (<5 nm from surface),10-12 and/or enhanced fluorescence of chromophores at a distance >10 nm.10
These properties are potentially useful in a broad range of applications, which range from photonic devices,11 biosensors,2, 13-15 nanoreactors5 to light harvesting devices.16 The precise size, shape and degree of aggregation of AuNPs has a significant effect on the nature of the properties listed above.1 However, citrate stabilized AuNPs, or those stabilized with small molecules, can readily aggregate to such an extent that these properties are lost. Stablisation of nanoparticles with polymers is one avenue to preservation of these properties and introduction of further functionality.
We have developed methods for stabilisation of gold nanoparticles with polymers synthesized by RAFT polymerisation17-19 and also investigated the mode of binding of phenyl dithioesters (a common class of RAFT agent) to gold nanoparticles.20 Figure 1 shows the process of generation of polymer stabilized gold nanoparticles where a) the polymer is PMMA a water insoluble The advantage of using RAFT polymers is that the thiocarbonylthio group is available at one end for attachment to the gold nanoparticle and it is possible to introduce a range of functionalities into the pendant groups or other end-group for derivatisation with functional molecules.

Figure 1 Photographs showing a) coprecipitation of AuNPs with PMMA end-functionalised with a thiocarbonylthio group and phase transfer of the polymer stablised gold nanoparticles into chloroform, and b) AuNPs before and after stabilisation with a water soluble poly PEG methacrylate with a thiocarbonyl thio endgroup, where no change in the SPR is observed but a change in size by DLS is observed after stabilisation.
One major line of research has been encoding of these polymer stabilized gold nanoparticles with molecular barcodes.18, 19 The detection mode used was surface enhanced Raman spectroscopy (SERS), where the high electric field that occurs at the surface of nanoparticles or between aggregates results in signal intensities that can be comparable to fluorescence. SERS has the added advantage of not being susceptible to photobleaching and the large number of narrow lines gives rise to greater potential for multiplexing (See Figure 2).
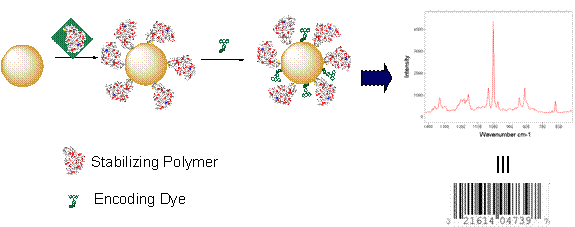
Figure 2 Schematic showing self assembly of stabilising polymers and SERS molecular barcodes around gold nanoparticles as well as an example of a SERS spectrum.
References
1. Glomm, W. R. J. Dispersion Sci. Technol. 2005, 26, 389-414.
2. Mulvaney, S. P.; Musick, M. D.; Keating, C. D.; Natan, M. J. Langmuir 2003, 19, 4784-4790.
3. Suzuki, M.; Niidome, Y.; Kuwahara, Y.; Terasaki, N.; Inoue, K.; Yamada, S. J. Phys. Chem. B 2004, 108, 11660-11665.
4. Joo, S. W.; Chung, T. D.; Jang, W. C.; Gong, M.-s.; Geum, N.; Kim, K. Langmuir 2002, 18, 8813-8816.
5. Manea, F.; Houillon, F. B.; Pasquato, L.; Scrimin, P. Angew. Chem., Int. Ed. 2004, 43, 6165-6169.
6. Budroni, G.; Corma, A. Angew. Chem., Int. Ed.2006, 45, 3328-3331.
7. Zhang, Z.-F.; Cui, H.; Lai, C.-Z.; Liu, L.-J. Anal. Chem. 2005, 77, 3324-3329.
8. Lioubashevski, O.; Chegel, V. I.; Patolsky, F.; Katz, E.; Willner, I. J. Am. Chem. Soc. 2004, 126, 7133-7143.
9. Pasquato, L.; Rancan, F.; Scrimin, P.; Mancin, F.; Frigeri, C. Chem. Comm. 2000, 2253-2254.
10. Eustis, S.; El-Sayed, M. A. Chem. Soc. Rev.2006, 35, 209-217.
11. Thomas, K. G.; Kamat, P. V. Acc. Chem. Res. 2003, 36, 888-898.
12. Schneider, G.; Decher, G. Nano Lett. 2006, 6, 530-536.
13. Stoeva, S. I.; Lee, J.-S.; Thaxton, C. S.; Mirkin, C. A. Angew. Chem., Int. Ed. 2006, 45, 3303-3306.
14. Talley, C. E.; Jusinski, L.; Hollars, C. W.; Lane, S. M.; Huser, T. Anal. Chem. 2004, 76, 7064-7068.
15. Rosi, N. L.; Mirkin, C. A. Chem. Rev. (Washington, DC, U. S.) 2005, 105, 1547-1562.
16. Kamat, P. V. J. Phys. Chem. B 2002, 106, 7729-7744.
17. Merican, Z.; Schiller, T.; Fredericks, P. M.; Blakey, I. Proceedings of ICONN '06. International Conference on Nanoscience and Nanotechnology. 2006, 622-625.
18. Merican, Z.; Schiller, T. L.; Hawker, C. J.; Fredericks, P. M.; Blakey, I. Langmuir 2007, 23, 10539-10545.
19.Schiller, T. L.; Fredericks, P. M.; Merican, Z.; Blakey, I. Proceedings of ICONN '06. International Conference on Nanoscience and Nanotechnology. 2006, 626-629.
20. Blakey, I.; Schiller, T. L.; Merican, Z.; Fredericks, P. M. Langmuir 2010, 26, 692-701.
Point of contact: Idriss Blakey
